
About 100 species of mammals are endemic to the southeastern United States, an area of diverse habitat types and high biodiversity. Many of these species are either formally considered aquatic or semi-aquatic, or they are otherwise closely associated with aquatic ecosystems. In the southeastern United States, greater than 80 percent of mammalian species are listed in some category of concern throughout all or at least a portion of their ranges (i.e., endangered, threatened, under review for possible listing as endangered or threatened, species of special concern, species deemed in need of management, etc.). Almost all of these species are imperiled because their populations have declined to dangerously low levels due to habitat loss, habitat degradation, or other human-related factors. Most imperiled mammal taxa can be categorized as belonging to one of two general groups: habitat generalists or habitat specialists. Typically, habitat generalists have become imperiled mainly due to human exploitation, while habitat specialists have suffered primarily from habitat loss or habitat degradation.
The passage of the U.S. Endangered Species Act of 1973 resulted in an increased need for information concerning distribution and status of all native species. However, relatively little is known concerning the historical distribution and current status of many mammalian taxa, and this is particularly so for small non-game species. In this chapter we provide species accounts of mammals commonly associated with aquatic ecosystems that we consider to be imperiled in the southeastern United States. In these accounts we have included information which we feel is valuable toward best understanding the threats that challenge each considered taxon.
The endangered West Indian manatee inhabits shallow coastal waters, estuaries, and rivers along the coast of the southeastern United States, throughout the Caribbean region, along the eastern coast of Central America, and on the northeastern coast of South America to Brazil (Odell, 1982). Florida is essentially the northern extent of its range, although individuals have occasionally been reported as far north as New Jersey on the Atlantic coast and as far west as Texas along the Gulf of Mexico coast (U.S. Fish and Wildlife Service, 1978). These extremely docile aquatic herbivores have a very low reproductive potential, and females are thought to give birth to a single calf at three- to five-year intervals. Longevity in the wild may reach 40-50 years. Manatees spend a major portion of their time feeding, and they may consume 10 to 15 percent of their body weight daily (Odell, 1982).
Historically, populations of West Indian manatees were severely reduced by hunting for meat, bone, hides, and fat (Bertram and Bertram, 1973; Peterson, 1974). More recently, they have suffered because of pollution, flood-control dams, and injuries from accidents with pleasure boats and barges. In Florida, collisions with pleasure boats appear to be the major identifiable cause of manatee mortality (Hartman, 1971). A number of other factors related to rapidly increasing coastal development appear to threaten manatee populations. Censusing these large mammals is surprisingly difficult. In 1985, however, at least 1,200 West Indian manatees were estimated to winter in Florida (Reynolds and Wilcox, 1986; OShea, 1988), where most of the United States population congregates.
The endangered Florida panther formerly occurred throughout the southeastern United States from central South Carolina through Tennessee, to northwestern Arkansas and south to the Gulf of Mexico. During the 1970s, the Florida panther was thought to be extinct, along with the remainder of the other subspecies of pumas in the eastern United States. During that decade, however, the existence of a population in and around Big Cypress National Preserve in southern Florida was confirmed.
The Florida panther is not restricted to aquatic ecosystems, but because of human pressures, this subspecies now survives only in habitats consisting of sawgrass prairies, cypress and oak hammocks, and permanently flooded wetlands. The density of white-tailed deer is relatively low in these habitats, and in these places panthers subsist primarily on feral hogs. Florida panthers require large home ranges: up to 260 km2 (100 square miles) for females and 1,040 km2 (approximately 400 square miles) for males (Maehr, 1990). Because these panthers move extensively, they are extremely sensitive to habitat fragmentation (Harris, 1985). The current range of the Florida panther encompasses some 800,000 ha (1,976,773 acres), and includes public lands of a national wildlife refuge, a national park and national preserve, a state preserve, a wildlife management area, and a Seminole reservation. About 50 percent of the Florida panthers range is on private land (Maehr, 1992a). Only 30-50 Florida panthers survive in the wild.
This semi-aquatic carnivore is classified as an Appendix II species under the Convention of International Trade in Endangered Species of Flora and Fauna. Until a few years ago, this classification was intended only for species that could become threatened if international trade was not strictly regulated. Today, the river otter is listed in Appendix II due to its status as a "look-alike" for pelts of endangered species of otters from South America and Africa.
The river otter was once abundant throughout most of the United States and was an important furbearer to colonial trappers (Schorger, 1970). Populations of river otters have been extirpated from much of their original range, partly due to indiscriminate, unregulated trapping (Godman, 1826; Flower and Lydekker, 1891; Duplaix and Simon, 1976) and losses of habitat (Park, 1971; Fimreite and Reynolds, 1973). During the 1950s and 1960s, not only was the quantity of suitable habitat diminished for river otters, but the quality was as well, due to the extensive use of DDT, heptachlor, dieldrin, and certain heavy metals (Clark et al., 1981). It is likely that bioaccumulation of those pollutants in this semi-aquatic species resulted in lowered reproduction or decreased survival. By the mid-20th century, river otters were common only in coastal regions of the southeastern United States.
The Everglades mink, under review for listing as endangered or threatened, is an isolated population of mink and is considered common in portions of the Everglades and Big Cypress National Park (Humphrey, 1992b). Water-control projects, resulting in changes in water levels, saltwater intrusion into the aquifer, altered fire regimes, and oxidation of peat soils, likely have been detrimental to this subspecies. Destruction of natural habitat to increase citrus production appears to be the greatest current threat to the Everglades mink. However, several major habitat tracts suitable to this mink are protected through federal ownership.
Major concentrations of Florida black bears occur in and around the Okefenokee National Wildlife Refuge in southern Georgia; Apalachicola National Forest, Osceola National Forest, and Ocala National Forest in the northern portion of Florida; and Big Cypress National Preserve in southern Florida. Estimates of current populations range from 500 to 1,000 individuals. Populations of black bears in Florida are probably the most fragmented in North America, and although the larger populations appear stable, extirpation of the smaller, more isolated populations probably will continue (Maehr, 1992b). Loss of suitable habitat, including cypress and hardwood swamps, represents the major threat to Florida black bears. Poaching and mortality caused by accidents with roadway traffic are also important factors in fringe populations due to this subspecies low reproductive and recruitment rates (Maehr, 1992b).
Historically, the Louisiana black bear was abundant in the lower Mississippi delta. However, due to the extensive loss of bottomland hardwoods caused by land clearing and human exploitation, the range of this subspecies has been severely restricted. Black bear populations are known to exist in the Atchafalaya River basin and Tensas National Wildlife Refuge in Louisiana. However, an introduction of U. a. americanus from Minnesota into Louisiana during the 1960s prompted debate among biologists over whether or not the Louisiana subspecies currently exists in its historic form.
The most important threat to the Louisiana black bear is continued loss of its habitat, although bear deaths caused by poaching and automobile collisions have also been documented and may represent significant mortality factors (Weaver, 1992). Populations have been estimated at greater than 60 bears in the Tensas River basin and greater than 30 bears in the Atchafalaya basin (Weaver, 1992).
The endangered Key deer historically occurred only at low densities in the Florida Keys, due to the dominance of mature forested lands. It is likely that hurricanes were a major influence on populations of Key deer, by operating to periodically create earlier successional-stage vegetation which was beneficial to this subspecies (Hardin et al., 1984).
The hunting of deer reduced the Key deer population to near extinction until it was banned by the Florida Legislature in 1939. The Key Deer National Wildlife Refuge was established in the early 1950s and a protection plan was developed. The Key deer population was estimated to be between 200 and 250 individuals in 1974 on Big Pine Key, with 100 to 150 deer estimated to live on 22 other keys (Klimstra et al., 1974). The overall population has apparently declined since that time (Hardin et al., 1984), and human-related destruction of deer habitat is no doubt an important factor responsible for this decline. However, the primary factor threatening the Key deer is mortality from collisions with automobiles (Klimstra et al., 1974; Klimstra, 1985), accounting for about 80 percent of all deaths.
The endangered lower keys marsh rabbit is present on several of the lower Flordia Keys (Howe, 1988). Recent records include Big Pine, Hopkins, Sugarloaf, Welles, Saddlebunch, Geiger, and Boca Chica keys, and remaining populations are highly fragmented (Wolfe, 1992). The lower keys marsh rabbit has apparently been extirpated from several other keys. This subspecies is also commonly known as the "Playboy bunny" since its original description was partially funded by the Playboy Foundation (Wolfe, 1992).
The lower keys marsh rabbit prefers marshes and adjacent low vegetative cover, especially grasses and sedges. However, it is sometimes found in more upland areas such as grassy fields and tropical hammocks (Wolfe, 1992). The greatest threat to this rabbit has been the destruction of its habitat for use by humans. Highway traffic and predation by domestic and feral pets also have caused extensive mortality.
This muskrat species of special concern is restricted to Florida and southeastern Georgia (Wassmer and Wolfe, 1983). Preferred habitat is shallow marshes with emergent vegetation such as maidencane and pickerelweed (Lefebvre and Tilmant, 1993). By far the greatest threat to the round-tailed muskrat has been the extensive destruction of its original wetland habitat. However, fragmentation of remaining habitat no doubt has also played a role in the demise of this species, as isolated populations are extremely vulnerable to destruction by fluctuations in water levels.
The endangered lower keys rice rat (or marsh rice rat) is widespread in the lower Florida Keys, but apparently absent from the upper and middle Florida Keys (Schwartz, 1952; Goodyear, 1987). This rats primary habitat is the upland-to-marine interface (Goodyear, 1987), and it is most abundant in tidal marshes (Wolfe, 1985, 1990). The primary threat to the lower keys rice rat is the destruction and fragmentation of its habitat for human development.
The endangered Key Largo woodrat is endemic to Key Largo, Florida (Sherman, 1955; Schwartz and Odum, 1957). This woodrat is primarily found in deciduous forest habitats and usually is absent from deforested and oldfield areas. The primary threat to the Key Largo woodrat is the destruction of its forest habitat for human developments (Humphrey, 1992a). In the early 1980s, over 4,000 housing units were under construction or approved for construction on Key Largo, and certainly some of this construction impacted this woodrat.
The endangered Florida saltmarsh vole is known only from Island Field Marsh along the shore of Waccasassa Bay, Levy County, Florida. It appears to be restricted to areas near the edge of patches of black rush and in patches of seashore saltgrass (Woods, 1992). The main threat to the survival of this vole is flooding of habitat during catastrophic weather such as hurricanes. Destruction and fragmentation of habitat by coastal development have also posed threats.
The endangered Key Largo cotton mouse is restricted to the northern half of Key Largo, Florida (Barbour and Humphrey, 1982). This species preferred habitat is deciduous forest in the dry-tropical-forest life zone (Holdridge, 1967). Human-related development and the resulting destruction and fragmentation of forest habitat has been the main threat to the survival of this mouse.
The endangered Alabama beach mouse occurs only along the coast in Alabama, where its habitat consists of sand dunes along beaches. Destruction and fragmentation of this habitat by human development has been the greatest threat to this mouses survival. Between 1921 and 1983, commercial and residential developments and human recreational activities destroyed over 60 percent of the habitat of this beach mouse (Holliman, 1983). Tropical storms also have resulted in mortality and habitat destruction.
The endangered Anastasia Island beach mouse occurs only on Anastasia Island, Florida. Substantial populations remain at each end of the island, with small populations scattered along its length (Humphrey et al., 1987). These mice are restricted to sand dunes vegetated mainly by sea oats and dune panic grass, and also to the adjoining scrub, characterized by oaks, sand pine, and palmetto. Habitat loss and fragmentation caused by human developments have been primary factors in the demise of this subspecies. Predation by domestic house cats and competition from house mice, which have colonized the dunes, also have had a detrimental impact on this mouse.
The endangered Choctawhatchee beach mouse is endemic to coastal dunes and historically was present as almost a continuous population along the coastal dunes between the entrances to Choctawhatchee and Saint Andrew bays, Florida. Currently, only two, or possibly three, small disjunct populations survive (Holler, 1992a). Optimal habitat consists of the primary and secondary dunes vegetated by sea oats, beach grass, and bluestem. Habitat loss caused by real estate development has been the single most important factor endangering this mouse subspecies (Bowen, 1968).
The endangered Perdido Key beach mouse is endemic to the coastal dunes between Perdido Bay, Alabama and Pensacola Bay, Florida. It currently occupies only 1.9 km (1.2 miles) of habitat at Gulf State Park, Alabama and 11 km (6.8 miles) of habitat at Gulf Islands National Seashore, Florida. It is considered the most endangered of the five endangered subspecies of beach mice (Holler, 1992b). Loss of habitat to real estate development and storm damage has resulted in the endangered status of this mouse. Vulnerability of remaining habitat and populations to storm damage is the greatest present threat to this subspecies survival (Holler, 1992b).
The threatened southeastern beach mouse is endemic to coastal dunes from south of Ponce Inlet in Volusia County to Hollywood Beach in Broward County, Florida. This subspecies principal habitat is the sea oats zone of coastal dunes, but grassland and open sandy areas with scattered shrubs are also occupied in some locations. Cumulative effects of real estate development, house cats, and house mice appear to render otherwise suitable habitat uninhabitable to this mouse, and in addition, erosion of beaches has destroyed significant mouse habitat (Stout, 1992).
The endangered Saint Andrew beach mouse is endemic to coastal dune habitat from the Saint Joseph spit in Gulf County to the entrance of Saint Andrew Bay in Bay County, Florida. The only substantial population remaining is in an 8 km (five mile) strip of high dunes at the northern end of the spit in Saint Joseph Peninsula State Park. A small population also exists on Crooked Island East in Bay County. Real estate development has destroyed and fragmented this subspecies habitat in many areas. Foot and vehicle traffic associated with recreational activities on sand dunes has resulted in serious erosion of the remaining mouse habitat (James, 1992).
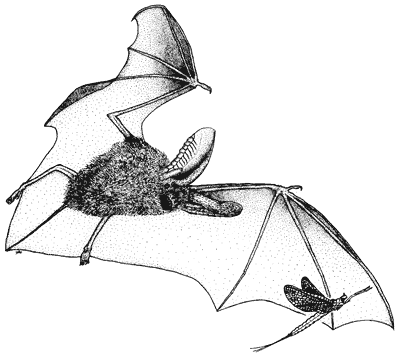
The endangered gray bat inhabits cave regions of Arkansas, Missouri, Kentucky, Tennessee, and Alabama, with occasional colonies and individuals found in adjacent states. Gray bats are cave residents year-round. Approximately 95 percent of all gray bats hibernate in only eight caves; two in Tennessee, three in Missouri, and one each in Kentucky, Alabama, and Arkansas (Harvey, 1992).
During summer, female gray bats form maternity colonies of a few hundred to thousands of individuals, often in caves containing streams. Male gray bats, along with non-reproductive females, form summer bachelor colonies in caves. Summer caves, especially those occupied by maternity colonies, rarely are located greater than 3 km (about two miles) from rivers or lakes. Each summer colony occupies a home range that often contains several roosting caves scattered along as much as 80 km (50 miles) of river or lake shore. Gray bats forage for insects primarily over water. Mayflies are apparently a major part of the diet, but like most bat species, they often feed on other insects as well.
Gray bat populations have suffered from habitat destruction, vandalism, disturbance to hibernation and maternity colonies by humans, pesticides, and other chemical toxicants. Population declines have been estimated at 89 percent in Kentucky, 72 to 81 percent in Missouri, 61 percent in Arkansas, and 76 percent in Tennessee and Alabama (Tuttle, 1979).
The endangered Indiana bat occurs in the eastern United States from Oklahoma, Iowa, and Wisconsin east to Vermont, and south to northwestern Florida. Distribution is associated with major cave regions and areas north of cave regions. About 85 percent of the remaining Indiana bat population hibernates at only seven locations: two caves and a mine in Missouri, two caves in Indiana, and two caves in Kentucky (Harvey, 1992). During summer, female Indiana bats form small maternity colonies of at most 100 individuals, usually under loose tree bark, primarily in wooded streamside habitat. Indiana bats usually forage for insects near foliage of streamside and floodplain trees.
Populations of Indiana bats continue to decline in size in spite of recovery efforts. Destruction of foraging and roosting habitat, vandalism, disturbance to hibernating colonies, and chemical toxicants have all likely played a role in this decline. Population estimates at major cave hibernacula indicated a 34 percent decline in numbers of individuals from 1983 to 1989 (Harvey, 1992).
This semi-aquatic mole occurs in the southeastern United States only in the Appalachian Mountains and along the Atlantic coast as far south as Georgia. It inhabits damp or muddy soil. Some tunnels constructed by this mole lead directly into water, and this species is an excellent swimmer. Food consists of aquatic insects, crustaceans, small fish, and annelids (van Zyll de Jong, 1983). The star-nosed mole can be considered imperiled in the southeastern United States because of its limited distribution and restrictive habitat type.
The threatened Dismal Swamp southeastern shrew occurs only in the Dismal Swamp National Wildlife Refuge in southeastern Virginia and adjacent portions of the swamp in North Carolina (French, 1980). The primary threat to this shrew has been the destruction of its swamp habitat, which is more accurately described as a wooded peat bog. The original Dismal Swamp has been reduced in size by roughly 85 percent since about 1900.
This semi-aquatic shrew occurs in the southeastern United States only at higher elevations in the Appalachian Mountains. The water shrew is an excellent swimmer and feeds primarily on aquatic insects, other aquatic invertebrates, and small fish (van Zyll de Jong, 1983). This shrew can be considered imperiled in the southeastern United States because of its very limited distribution.
The aquatic, semi-aquatic, or otherwise aquatic habitat dependent mammal taxa discussed in this paper are considered imperiled for a number of reasons, almost all of which are human related. Destruction, alteration, and fragmentation of habitat, along with other forms of environmental degradation, are no doubt the foremost causes of imperilment (summarized in Table 1). Quite likely, some of these taxa will be extirpated in the foreseeable future unless immediate steps are taken to arrest or reverse current trends. The question is, "beach houses or beach mouses?"
Table 1. Summary of imperiled aquatic, semi-aquatic, and otherwise aquatic habitat associated mammal taxa of the southeastern United States, their level of imperilment, and the primary reasons for their imperilment. |
||||
Order:Family: |
Species/SubspeciesCommon Name 1 |
Aquatic Association:Regional Location |
Level of Imperilment |
Primary Causeof Imperilment |
Sirenia: |
||||
|
West Indian manatee |
Aquatic: southeast U.S. coast, estuaries & rivers |
Endangered |
Habitat loss & alteration, hunting, pollution, boat traffic related mortalities |
Carnivora: |
||||
|
Florida panther |
Closely associated: FL |
Endangered |
Habitat loss & fragmentation, road traffic related mortalities |
|
river otter |
Semi-aquatic: throughout southeastern U.S. |
Threatened locally |
Habitat loss, trapping,pollution 2 |
Everglades mink |
Semi-aquatic: FL |
Threatened locally |
Habitat loss & alteration,pollution |
|
|
Florida black bear |
Closely associated: FL & GA |
Threatened locally |
Habitat loss, poaching, road traffic related mortalities |
Louisiana black bear |
Closely associated: LA |
Threatened locally |
Habitat loss, poaching |
|
Artiodactyla: |
||||
|
Key deer |
Closely associated: FL |
Endangered |
Habitat loss, hunting,road traffic related mortalities |
Lagomorpha: |
||||
|
lower keys marsh rabbit |
Closely associated: FL |
Endangered |
Habitat loss, road traffic related mortalities, predation caused by pets |
Rodentia: |
||||
|
round-tailed muskrat |
Semi-aquatic: FL & GA |
Threatened locally |
Habitat loss & alteration |
lower keys rice rat |
Closely associated: FL |
Endangered |
Habitat loss & fragmentation |
|
Key Largo woodrat |
Closely associated: FL |
Endangered |
Habitat loss & fragmentation |
|
Florida saltmarsh vole |
Closely associated: FL |
Endangered |
Habitat loss & fragmentation |
|
Key Largo cotton mouse |
Closely associated: FL |
Endangered |
Habitat loss & fragmentation |
|
Alabama beach mouse |
Closely associated: AL |
Endangered |
Habitat loss & fragmentation, storm related habitat damage |
|
Anastasia Island beach mouse |
Closely associated: FL |
Endangered |
Habitat loss & fragmentation, predation caused by pets |
|
Choctawhatchee beach mouse |
Closely associated: FL |
Endangered |
Habitat loss & fragmentation |
|
Perdido Key beach mouse |
Closely associated: AL & FL |
Endangered |
Habitat loss & fragmentation, storm related habitat damage |
|
southeastern beach mouse |
Closely associated: FL |
Threatened |
Habitat loss & fragmentation, storm related habitat damage, beach erosion |
|
Saint Andrew beach mouse |
Closely associated: FL |
Endangered |
Habitat loss & fragmentation, beach erosion |
|
Chiroptera: |
||||
|
Indiana bat |
Closely associated: southeastern U.S. |
Endangered |
Habitat loss, human disturbance, vandalism, chemical toxicants |
gray bat |
Closely associated: southeastern U.S. |
Endangered |
Habitat loss, human disturbance, vandalism, chemical toxicants |
|
Insectivora: |
||||
|
star-nosed mole |
Semi-aquatic: Appalachian Mtns. & Atlantic coastal states |
Threatened locally |
Habitat loss, limited distribution |
|
Dismal Swamp southeastern shrew |
Closely associated: VA & NC |
Threatened |
Habitat loss |
water shrew |
Semi-aquatic: Appalachian states |
Threatened locally |
Habitat loss,limited distribution |
|
1 For scientific names refer to text. |
||||
2 As explained in text, part of the reason this species is regulated is due to the fact that it looks like some other imperiled otter species. |
||||